In This Section
- Understanding acids and bases
- The effects of positive and negative ions
- Different soils for different plants
- Finding the materials you'll need
- Guarding against contamination
- Considering other botany projects
Although this section is classified as a botany project, you'll notice as you work through it that it contains a fair amount of chemistry, as well.
It's not unusual for scientific areas, or disciplines, to cross over in the course of a project or experiments. This project is a good example of how that occurs.
You've probably heard of the pH scale, or heard someone talk about the pH factor of a particular material. But what is pH exactly, and how does it affect the growth of plants?
In this section, we'll explore the basics of pH, and experiment to learn how the pH factor of liquid affects the germination and growth of bean seeds. By the time you finish, you'll have had valuable lessons in both botany and chemistry, and have a better understanding of how branches of science overlap.
So What Seems to Be the Problem?
You know that plants need certain things to help them grow. They need some kind of growing medium, usually dirt. They need light, and they need water.
The problem you'll attempt to solve while doing this science fair project is whether the pH of the water with which plants are sprinkled affects the rate of growth.
To get a better idea of what you'll be doing, and to help you formulate a hypothesis, it's important that you have a general understanding of exactly what pH is.
The initials pH stand for percent hydronium ion. The pH scale is used as a measure of how acidic or basic a liquid is. But how do liquids become acidic or basic? Isn't a liquid just a liquid?
Water-and distilled water, at that-is the only liquid that is neutral. That means it's right in the middle of being acidic or basic-and it's neither. It's just pure water.
The pH scale starts at zero and ends at 14. The more acidic a liquid is, the lower its number on the pH scale. The less acidic-or more basic a liquid is-the higher its number.
Most of the liquids you encounter on a daily basis are just around neutral. They might be a little above or a little below, but most liquids tend to be closer to neutral than at either end of the pH scale.
Liquids get their pH level as a result of molecules that split apart to form positive and negative ions. An ion is the loss or gain of electrons from an atom. When an atom loses electrons, it forms a positive ion. When an atom gains electrons, it forms a negative ion.
Liquids will be either acidic or basic (also called alkaline), depending on whether they contain positive or negative ions. If there are more positive ions in the water, the water is more acidic. If there are more negative ions in the water, the liquid is more basic.
In this experiment, you'll control the pH of the water you'll use on bean plants by adding certain substances to make distilled water either acidic or basic. You'll also control all other factors, such as how much water and light each plant gets.
If you want to, you can use the name of this section, "Does the pH of Water Affect the Growth of Bean Plants?" as the title for your project. Other names to consider might be:
- What Type of Soil Do Bean Plants Prefer?
- Acid or Alkaline-What's Right for Bean Plants?
- To Grow or Not to Grow: Acidic vs. Alkaline Soil for Bean Plants
When you've finished with the experiment, you'll know whether bean plants prefer water that is acidic or basic.
What's the Point?
Some plants prefer acidic conditions. We call these acid-loving plants. Acid-loving plants include the following:
- Holly, pine, fir, spruce, birch, oak, magnolia, willow, and flowering crabapple trees
- Rhododendrons and azaleas
- Hydrangeas
- Roses
- Cranberry, strawberry and blueberry plants
- Mountain laurel
- Crocuses
Other plants, however, such as the ones listed here, prefer alkaline soil:
- Yew, boxwood, and barberry shrubs
- Flowering plum and cherry trees
- Ash, beech, filbert, and maple trees
- Clematis
- Mock orange
- Lilacs
- Pinks
- Asters
Gardeners often help plants along by making the soil in which they grow either more acidic or more alkaline. There are products available, such as Miracid, that boost the acidity of soil. Garden lime (its chemical name is calcium carbonate) will help make soil alkaline.
In the experiment described below, you'll use distilled water as your control, and water with varying pH levels as your variables. This will allow you to observe the effects that liquids of varying pH levels have on the bean plants.
Who knows? You may end up increasing your interest in, or developing an interest in, gardening through this project. If nothing else, it will give you a better understanding of how plants grow and what types of factors affect them.
What Do You Think Will Happen?
If you've had experience with gardening and growing plants, you know that plants react differently to all sorts of factors.
Some plants like to be watered frequently, while others like to wait for a drink until the soil is completely dry. Some are much more susceptible to heat or cold than others. Some plants thrive in sunlight, while others like shady conditions. You've already read that some plants prefer an acidic soil, while others like basic soil.
You can do some research about growing bean plants to help you form a hypothesis. If you don't have much experience with plants, or don't have a good understanding of pH, it probably would be beneficial for you to learn more about growing plants, different types of soil, and so forth.
Or you can simply consider what you may already know about growing plants and make an educated guess about what will happen to the bean plants with which you'll be working.
Materials You'll Need for This Project
One thing about this experiment is that it's going to take some advance planning and a significant amount of time. You'll need almost a month from the time you plant the seeds until the time you draw final conclusions about the growth of the plants.
You also will need a material or two with which you may not be familiar. Most of the materials you'll need though, are common household items. You'll need:
- A substance used to adjust the pH level of water. We suggest a set of products called pH Up and pH Down, a brand that's readily available in pet supply stores. You'll need a bottle of each pH Up and pH Down. Retail cost is about $3.50 a bottle.
- pH test strips or test kit. You also can purchase these supplies from your local pet supply store, hobby shop, or from online sources. A package of test strips in a hobby shop should run you somewhere about $3 or $4. You should have 50 strips to make sure you have enough for the experiment. Or, your science teacher may have extra test strips that you could get for this project. It doesn't hurt to ask, right?
- Seven large-size plastic drinking cups. Cups should be about 16 ounces to allow room for bean plants to grow.
- Soil to fill the cups about three-quarters full. You'll need to use the same soil for all the cups. Buying a bag of potting soil is recommended.
- Twenty-one bean seeds
- Distilled water
- Metric ruler
- Seven two-liter, plastic bottles, empty and washed well
- Small paper cups in which to measure water
If you're going to order supplies from an online provider, be sure to do so ahead of time so you don't get stuck, unable to begin your project.
Conducting Your Experiment
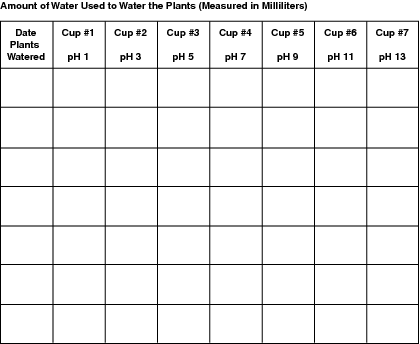
It's very important that the cups containing the bean seeds are all kept in the same conditions. They each need to have the same amount of light, heat, and so forth.
And it's extremely important that each plant receives the same amount of water. If you give the plants different amounts of water, you'll be unable to determine whether the plant was affected by the pH of the water, or simply the varying amount.
You can't mix water from any of the two-liter bottles, or use the same container to hold water from different bottles without washing it out in between. That's why it's recommended that when you water plants, you use small paper cups, pouring water from each two-liter bottle into its own paper cup, and then onto the bean plant.
If your plants don't need a full cup of water, measure up to the halfway mark of each cup and make a line. Fill the cup with water up to the line to assure that each plant gets the same amount.
Follow these steps to conduct the experiment:
1. Starting with seven two-liter bottles of distilled water, prepare each bottle so it has a specific pH value. Leave one bottle untreated (your control), with a pH level of 7. Add pH Up or pH Down to the other bottles so that one bottle has a pH level of 1, one has a pH level of 3, one has a pH level of 5, one has a pH level of 7, one has a pH level of 9, one has a pH level of 11, one has a pH level of 13. You'll raise or lower the pH level for each bottle from 7, depending on whether you're making the water acidic or alkaline. Cap the bottles tightly, label each one so you know which is which, and place them in an undisturbed location.
Do write down how many drops of either pH Up or pH Down you need to add to each two liter bottle. If you need to make more water at the different pH levels, you'll already know how much of the pH product to add, and the second round of water will have the exact same pH levels as the first round.
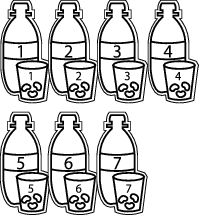
2. Plant three bean seeds into each of the seven large, plastic cups that have been filled about three-quarters of the way with potting soil.
3. Mark each cup and match one cup with one two-liter bottle of water. It's extremely important that each cup is watered from the same bottle each time, and not from any other bottle.
4. Making sure each cup gets the same amount of water, water the seeds in each cup so that the soil is moist, but not saturated. You might want to transfer water from the two-liter bottle to the cup with a tablespoon, allowing you to exactly measure how much each pot will get. Just be sure to wash the tablespoon between using it to handle water from different bottles.
5. Observe the cups every day, watering when the soil appears dry. Just be sure to always give the seeds in each cup the same amount of water. The plants will require more water as they get larger.
6. Using a metric ruler, measure the plants and record your observations every four days. Record the growth of the plants in a chart like the one found in the next section, "Keeping Track of Your Experiment."
Your experiment will be finished after 28 days, meaning you will have measured each plant seven times.
Keeping Track of Your Experiment
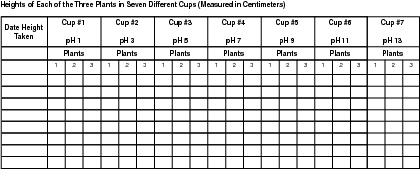
Make sure to note the height of each plant within the seven cups-21 plants in all-each time you measure. Keep track of how much water you've given the plants as well. The amount of water given to the plants in each cup should be the same.
Data charts to record water application and plant growth are provided, or you can make your own charts, if you prefer.
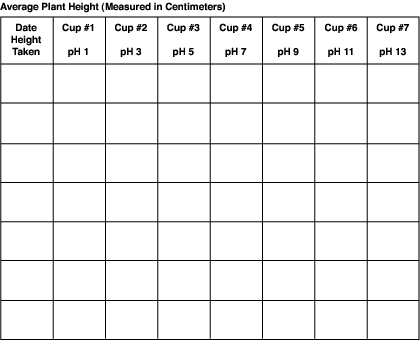
To calculate the average height for the three plants in each cup:
- Add the three heights together for each of the three plants in cup #1.
- Divide that total by 3.
- Record this number in data chart 2.
Repeat steps 1-3 for the remaining six cups.
Don't assume that all plants within a cup will grow equally. You can average the height of the three plants within each cup.
Putting It All Together
Use the information contained on your data charts to make some graphs, plotting the growth of each plant. Or, you can summarize your observations in written form, if you prefer.
Did some of the plants grow quickly at first, only to halt their growth later? Did any of the plants die? Did your hypothesis prove to be correct?
Look for trends and patterns, concluding which plants had the best overall growth, the fastest starts, and so forth.
Further Investigation
If you enjoyed this experiment and want to try another variation of it, you could try to grow plants hydroponically-that is, in water instead of dirt-while varying the pH level of the water.
It would be interesting to see if you get different results than you did when you used water of varying pH levels to water plants growing in dirt.
You also could put liquids other than water on plants to see how growth was affected. Or, you could work with acid-loving plants and base-loving plants, testing to see at what pH level they grow best.
Hands-on look at the "basics" of acids and alkalis
Introduce pupils to botany and the basics of acids and bases with this printable science activity. In this hands-on science project, pupils grow bean plants using water with various levels of pH.




